CareOne Lidocaine Patches Fail to Provide Advertised Pain Relief, Class Action Alleges
Last Updated on February 9, 2026
Fuller v. The Stop & Shop Supermarket Company LLC
Filed: November 17, 2022 ◆§ 7:22-cv-09824
Stop & Shop faces a class action that claims the packaging of its CareOne adhesive lidocaine patches deceptively overstates the product's effectiveness.
New York
The Stop & Shop Supermarket Company faces a proposed class action that claims the packaging of its CareOne adhesive lidocaine patches deceptively overstates the product’s effectiveness.
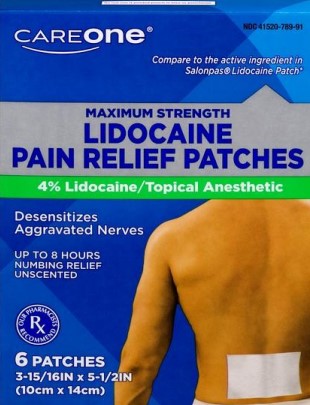
The 14-page lawsuit alleges that the topical anesthetic patches regularly peel off the body within only a few hours of being applied, despite Stop & Shop’s representation that the patches can provide “up to 8 hours” of “maximum strength” pain relief.
The case also argues that Stop & Shop’s claims that the patches can “Desensitize[] Aggravated Nerves” and provide hours of “numbing relief” lead consumers to wrongly assume that the product can perform medical treatments on par with prescription-strength, FDA-approved items, even though OneCare patches possess neither attribute.
Want to stay in the loop on class actions that matter to you? Sign up for ClassAction.org’s free weekly newsletter here.
“Adequate adhesion is critical for topical delivery systems, because if the patch lifts or detaches while walking, sleeping or exercising, dosing will be compromised,” the case states. “However, the Product cannot adhere to the skin for more than four hours, which renders the ‘Up To 8 Hours’ misleading, a significant disparity.”
Although newly developed adhesive technology can maintain adhesion to the skin for at least eight hours under normal conditions, CareOne’s product uses outdated and defective adhesion methods proven ineffective in a 2021 Journal of Pain Research study, the case says.
“The ‘Up To 8 Hours’ claim is also inconsistent with the ‘Uses’ disclosed on the Drug Facts which indicates it can only provide ‘Temporary relief’ which consumers will understand as a short time, less than eight hours,” the filing further argues.
Further still, the suit contends that CareOne patches cannot deliver the “maximum strength” amount of lidocaine as advertised due to the product’s systematic failure to adhere properly to the skin. The complaint charges that this label statement leads consumers to falsely believe that CareOne patches are “superior or equivalent in efficacy and results” to other non-defective, over-the-counter and prescription-strength lidocaine patches.
According to the case, packaging statements about desensitizing nerves and numbing pain wrongly imply that the product can “completely block and numb nerves and pain receptors, eliminate responses to painful stimuli, and treat neuropathic and musculoskeletal pain, including back and spinal pain.” These desensitizing and numbing front-label promises are “inconsistent with what it is authorized by law to say, i.e., that it can be used to ‘temporarily relieve[s] minor pain,’” the case states.
“As the Product’s ‘Uses’ purports to provide ‘temporary relief of pain,’ without qualifying the pain it purports to relieve as ‘minor,’ even consumers who scrutinize the Drug Facts on the back label will be misled,” according to the complaint.
Finally, the filing alleges a seal with the universally recognized symbol for a prescription, “Rx,” on the patches’ front label gives consumers the false impression that the over-the-counter product will be prescription strength.
The case argues that these misleading representations allow Stop & Shop to sell its lidocaine patches at a higher price.
The lawsuit looks to cover anyone in New York, New Jersey, New Hampshire or Rhode Island who purchased the CareOne adhesive lidocaine patches during the applicable statute of limitations period.
Get class action lawsuit news sent to your inbox – sign up for ClassAction.org’s free weekly newsletter here.
Video Game Addiction Lawsuits
If your child suffers from video game addiction — including Fortnite addiction or Roblox addiction — you may be able to take legal action. Gamers 18 to 22 may also qualify.
Learn more:Video Game Addiction Lawsuit
Kratom 7-OH Lawsuits
Anyone who has used 7-OH kratom products and suffered a serious injury, such as overdose, heart attack or addiction, may be able to take legal action.
Read more: Kratom 7-OH Lawsuits
How Do I Join a Class Action Lawsuit?
Did you know there's usually nothing you need to do to join, sign up for, or add your name to new class action lawsuits when they're initially filed?
Read more here: How Do I Join a Class Action Lawsuit?
Stay Current
Sign Up For
Our Newsletter
New cases and investigations, settlement deadlines, and news straight to your inbox.
Before commenting, please review our comment policy.