‘Wasteful’: Class Action Claims Genentech Reaped Millions by Selling Cancer, Asthma Drugs in Excessive Dosage Amounts
by Erin Shaak
Williamson et al. v. Genentech, Inc. et al.
Filed: September 24, 2020 ◆§ 3:20-cv-06695
A class action claims Genentech has sold medications in single-use vials that contain more medicine than is appropriate for most patients, causing excessive waste.
California
Genentech, Inc. and Genentech USA, Inc. face a proposed class action that claims the “wasteful way” in which the drug makers have packaged three cancer drugs and an asthma medication has caused patients and insurers to pay millions for medicines that end up “simply being thrown away.”
According to the case, the third amended complaint in the litigation, San Francisco-based Genentech has sold four expensive medications in single-use vials that contain more medicine than is appropriate for most patients, meaning a significant portion of each medication goes to waste each time a patient receives treatment.
Per the complaint, Genentech and other drug manufacturers are “finding a way to charge patients and insurers for drugs that they don’t even take,” with hundreds of millions of dollars being paid each year for unused medications “that go in the trash.”
The plaintiffs, a follicular lymphoma patient and his insurer, claim Genentech’s packaging of four medications—Avastin, a colorectal cancer treatment; Rituxan, used to treat Non-Hodgkin’s Lymphoma and other conditions; Kadcyla, a breast cancer treatment; and Xolair, which is used to treat asthma—has needlessly added to the already “crushing” costs of healthcare that many patients, especially those being treated for types of cancer, find difficult or impossible to afford.
“Cancer drugs have become so expensive that even middle-class patients have been forced to stop taking their medicines—at great risk to their survival—because they cannot afford them,” the suit reads.
Cited in the case is a 2016 BMJ (formerly the British Medical Journal) paper in which a group of experts headed by Dr. Peter B. Bach discovered a “systemic source of waste” that has come “at great cost” to consumers. Dr. Bach’s study, according to the suit, estimated that the wasted portions of 20 cancer drugs, including three manufactured by the defendants, that were thrown away as a result of larger-than-needed vial sizes would rake in $1.8 billion in revenues for pharmaceutical companies in the year 2016 alone.
The Bach paper found that if each manufacturer, including Genentech, would offer just one additional smaller vial size for each of the products in the study, consumers and their insurers would realize annual savings of $1.4 billion, not accounting for savings of hundreds of millions more in markups from doctors and hospitals, the lawsuit states.
Genentech, however, has failed to follow the recommendations in the 2016 study and continues to boost revenues by charging consumers for more medicine than they can use, the lawsuit argues, alleging violations of the California Unfair Competition Law.
The amount spent on wasted medication for just one patient can total “many thousands of dollars a year” for Genentech’s drugs, the case charges, providing the following example:
“Avastin is sold in vials containing either 100 mg or 400 mg. The initial dosage of Avastin for patients with lung cancer at the outset of treatment is 15 milligrams per kilogram of body weight, or 1,218 mg for the average male patient and 1,013.1 for the average female patient (average weight: 81.20 kg for men and 67.54 kg for women). To meet that dose, the average male patient must receive three 400 mg vials and one 100 mg vial (1,300 mg altogether), with 82 mg, or 82% of the 100 mg vial, being unused and thrown away. The average female patient would receive two 400 mg vials and three 100 mg vials (1,100 mg in total), with 86.1 mg, or 86.1% of the last 100 mg vial, being unused and going to waste.”
According to the case, the cost of the wasted Avastin in the example detailed above would have been $756.06 for the average male patient in February 2019 and $803.31 for the average female patient in May 2017. Since Genentech’s recommended treatment course for Avastin calls for the drug to be administered every three weeks, the company reaps $12,096.96 for each male patient and $12,852.96 for each female patient every year on medication that is thrown away, the suit claims, adding that patients end up paying even greater amounts due to wholesale and retail markups.
The lawsuit details similar examples for Rituxan, Kadcyla and Xolair, alleging that Genentech has generated millions of dollars in annual revenue from the unused portions of its medications.
Moreover, the case says asthma medication Xolair was sold only in 150 mg vials until 2018, even though the FDA approved the product in 75 mg vials. For many patients, their dose of Xolair is exactly 75 mg, 225 mg or 375 mg, meaning half of one vial “invariably had to be discarded” for each treatment, the lawsuit relays.
Per the complaint, Genentech could have “substantially reduced” the amount of wasted medication by adding just one additional vial size for each drug as the 2016 Bach paper recommended. The lawsuit includes the following chart to illustrate the alleged waste reduction percentage for the three cancer drugs after adding an additional vial size:
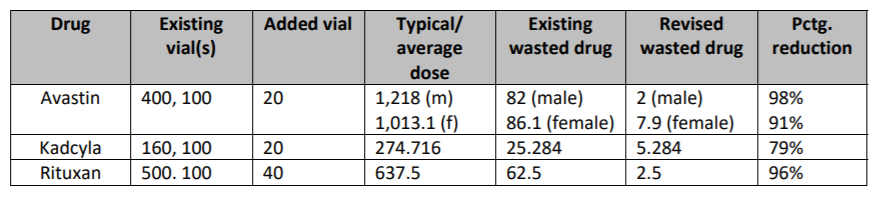
For Xolair, if Genentech had introduced a 75 mg vial before 2018 as it did in Europe, the company could have eliminated all waste of the drug, the suit says.
“There is no legitimate reason why, before late 2018, Genentech could not have given U.S. patients the benefit of the smaller vial sizes that it gave to patients in Europe,” the complaint scathes.
The plaintiff, who underwent three rounds of treatment for follicular lymphoma between January 2016 and March 2018, alleges that if Genentech had made available a 40 mg vial of Rituxan, he and his insurer could have saved nearly $10,000 in unused medication.
Get class action lawsuit news sent to your inbox – sign up for ClassAction.org’s newsletter here.
Video Game Addiction Lawsuits
If your child suffers from video game addiction — including Fortnite addiction or Roblox addiction — you may be able to take legal action. Gamers 18 to 22 may also qualify.
Learn more:Video Game Addiction Lawsuit
Kratom 7-OH Lawsuits
Anyone who has used 7-OH kratom products and suffered a serious injury, such as overdose, heart attack or addiction, may be able to take legal action.
Read more: Kratom 7-OH Lawsuits
How Do I Join a Class Action Lawsuit?
Did you know there's usually nothing you need to do to join, sign up for, or add your name to new class action lawsuits when they're initially filed?
Read more here: How Do I Join a Class Action Lawsuit?
Stay Current
Sign Up For
Our Newsletter
New cases and investigations, settlement deadlines, and news straight to your inbox.
Before commenting, please review our comment policy.