No Science Supports Six-Pack Ab, Fat Loss Claims for Slendetone’s Flex Belt, Class Action Alleges [UPDATE]
Last Updated on July 21, 2021
Loomis v. Slenderstone Distribution, Inc.
Filed: May 7, 2019 ◆§ 3:19cv854
Slendertone Distribution's Flex Belt is the subject of a class action that claims no science backs the company's claims that the product can give users six-pack abs or reduce belly fat.
Case Updates
October 7, 2020 – Lawsuit Settled for $175,000
The proposed false advertising class action detailed on this page has settled, with Flex Belt maker Slendertone Distribution agreeing to pay $175,000, according to a September 30 court filing.
The plaintiff’s motion for preliminary settlement approval states that the deal comes after more than a year of litigation and in light of “Slendertone’s assertion of numerous factual and legal defenses and the effect of the current COVID-19 pandemic” on the company’s business and operations. In addition to the settlement’s monetary component, Slendertone said it has changed its advertising and website to omit statements the court found may be misleading to consumers, according to the filing.
The proposed deal covers anyone in California who, between May 7, 2015 and the date the settlement receives preliminary approval, bought a Slendertone Flex Belt for personal or household use. Once the settlement receives preliminary approval, claims can be filed online through an official settlement website. The parties handling the settlement negotiations estimate the class includes roughly 20,000 California consumers.
To combat fraud, Flex Belt buyers will need to submit proof of purchase, such as a digital receipt, confirmation email or order number, with their claims. Depending on the claims rate, Flex Belt buyers can expect to receive a refund of 9.5 percent to 28 percent of the total purchase price.
ClassAction.org will update this page when the official settlement website is live.
Slendertone Distribution’s “Flex Belt” electrical muscle stimulator is the subject of a proposed class action lawsuit centered on the apparent lack of science to back claims that the product can give users six-pack abs and reduce belly fat.
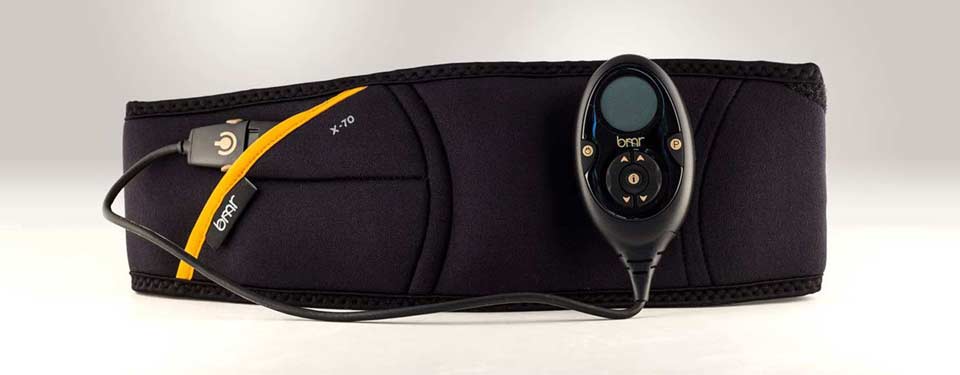 The Flex Belt is marketed and advertised by Slendertone as a “miracle device” that can give users “strong, toned abs” and “get rid of belly fat” with little to no exertion in just a few weeks, the suit states. According to the lawsuit, however, Slendertone’s advertising of the Flex Belt, which includes phrases such as “FDA-cleared technology,” falsely conveys the product’s supposed benefits – in particular, that it can work as a total replacement for traditional abdominal exercise and provide visible six-pack abs. Notwithstanding the product’s ostensible benefits, Slendertone also misleadingly claims the Flex Belt is “the first Ab Belt Toning system cleared by the FDA,” the case says.
The Flex Belt is marketed and advertised by Slendertone as a “miracle device” that can give users “strong, toned abs” and “get rid of belly fat” with little to no exertion in just a few weeks, the suit states. According to the lawsuit, however, Slendertone’s advertising of the Flex Belt, which includes phrases such as “FDA-cleared technology,” falsely conveys the product’s supposed benefits – in particular, that it can work as a total replacement for traditional abdominal exercise and provide visible six-pack abs. Notwithstanding the product’s ostensible benefits, Slendertone also misleadingly claims the Flex Belt is “the first Ab Belt Toning system cleared by the FDA,” the case says.
Though the case concedes that “limited science” does support that the Flex Belt can offer some strength and endurance benefits to abdominal muscle tissue, no science exists to support the claim that the product can wipe away belly fat, provide more attractive abs, or replace traditional exercise methods, according to the suit.
“Several other studies have resulted in similar findings and while some have concluded that [electronic muscle stimulation] can strengthen abdominal muscles or even improve endurance in abdominal muscles,” the complaint reads, “no study has ever yielded results supporting the notion that the use of EMS will result in body contouring, weight loss, visible results (including so-called ‘six-pack abs’), or that use can replace traditional exercise to improve overall health.”
Also noted in the lawsuit is the fact that the Flex Belt is considered a Class II Medical Device by the FDA and therefore requires pre-market approval before being sold. The FDA, the complaint says, has typically only approved devices like the Flex Belt that are claimed to “temporarily strengthen, tone or firm a muscle” while disapproving those that are claimed to aid with weight loss, help contour the body, develop toned abs or otherwise take the place of exercise. Proposed class members relied on Slendertone’s representations when purchasing the Flex Belt, which the lawsuit argues is worth less than its selling price.
Before commenting, please review our comment policy.